Recently, Jenscare Scientific Co., Ltd. ("Jenscare Scientific") successfully completed the first-in-human clinical study of its iJensRobo robot-assisted transcatheter tricuspid valve replacement (TTVR) system at Prince of Wales Hospital (PWH) in the Hong Kong Special Administrative Region.
This milestone not only marks a crucial step forward in Jenscare Scientific's integration of "interventional devices + surgical robotics", but also represents a breakthrough advancement of domestically developed high-end medical equipment in the field of complex structural heart disease interventions. It provides a new solution for the precise treatment of tricuspid valve disease worldwide.
A Powerful Collaboration:
Industry–Academia–Research–Clinical Synergy Tackles Key Challenges
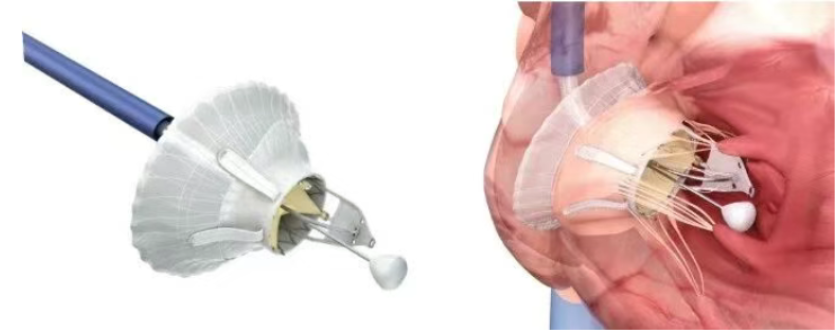
The LuX-Valve series—Jenscare Scientific's self-developed transcatheter replacement system for severe tricuspid regurgitation—overcomes numerous anatomical challenges associated with the condition through its innovative anchoring design, while significantly simplifying the implantation procedure. Meanwhile, the iJensRobo robotic-assisted interventional platform, developed through close collaboration between Jenscare Scientific and the Institute of Automation of the Chinese Academy of Sciences, is designed to deliver more precise, stable, and controllable support for TTVR procedures. By integrating high-precision motion control, active safety constraint mechanisms, and intelligent navigation technologies, the iJensRobo-assisted TTVR system enables stable and delicate device manipulation, enhancing the safety and reliability of critical procedural steps.
Building on the already user-friendly design of the LuX-Valve series, the addition of robotic technology further lowers the technical barrier, while significantly improving procedural accuracy and ease of use. This clinical study was also supported by the Hong Kong Innovation Institute of the Chinese Academy of Sciences and the Faculty of Medicine at The Chinese University of Hong Kong, forming a complete innovation loop from technology development to clinical translation.
Technological Breakthrough:
Four Core Advantages Redefining Precision Intervention
The iJensRobo system is not merely an auxiliary tool, but a robotic interventional platform specifically designed to address the complex anatomical scenarios encountered in TTVR procedures. Building on the inherent ease-of-use advantages of the LuX-Valve series compared to similar products, it further enhances precision and stability:
1) Precise control, eliminating human error
Minimally invasive interventional procedures are often affected by hand tremors and variations in operator experience. By combining the operational advantages of the LuX-Valve series with high-precision motion control and active safety constraints, iJensRobo filters out subtle hand tremors, making valve implantation more stable and precise. This reduces risks such as device deviation, operational errors, and instability in device navigation paths. It also decreases reliance on individual operator experience, helping extend the benefits of the LuX-Valve series to more patients worldwide.
2) Intelligent guidance, reducing dependence on experience
Traditional surgical tricuspid valve procedures are complex and high-risk. Existing minimally invasive transcatheter approaches on the market also require a high level of operator experience and imaging expertise. While the LuX-Valve Plus TTVR procedure already offers significant operational advantages due to its unique anchoring mechanism, iJensRobo further enhances it by providing structured guidance for critical steps through intelligent navigation. This enables procedures in complex anatomical scenarios to be performed under standardized and protocol-driven guidance, allowing more physicians to carry out the surgery safely.
3) Master-slave architecture, protecting physician health
The primary operator can perform the procedure remotely from a control console, significantly reducing radiation exposure and improving occupational health conditions.
4) Remote empowerment, overcoming spatial limitations
The system integrates remote operation capabilities, enabling cross-regional collaboration and training. In the future, it is expected to help extend access to high-quality medical resources to a broader patient population.
Product Synergy:
First Clinical Study Validates the Dual Advantages of "Interventional Devices + Robotics"
In this procedure, the iJensRobo system worked in seamless synergy with the LuX-Valve Plus Transvenous Tricuspid Valve Replacement System. As one of Jenscare Scientific’s core products, the LuX-Valve Plus is specifically designed for patients with severe tricuspid regurgitation. Its unique leaflet grasping plus interventricular septal anchoring mechanism offers significant clinical advantages in terms of safety and efficacy when addressing complex tricuspid valve anatomy.
In this clinical study, with the assistance of the iJensRobo robotic system, the LuX-Valve Plus valve was successfully implanted in the patient. Postoperatively, tricuspid regurgitation was completely eliminated immediately, with no paravalvular leak observed and excellent valve stability. These results fully validate the outstanding performance of the "interventional devices + surgical robotics" combination in complex TTVR procedures, laying a solid foundation for large-scale clinical application.
Strategic Significance:
Positioning at the Technological Frontier, Leading Industry Innovation
The successful completion of the first clinical case of the iJensRobo robot-assisted TTVR system represents a major achievement in Jenscare Scientific's dual-engine strategy of "interventional devices + surgical robotics" in the field of structural heart disease. The company has consistently focused on addressing unmet clinical needs by delivering high-quality innovative medical devices. Globally, there is a large population of patients suffering from severe tricuspid regurgitation who urgently require treatment. However, the adoption and accessibility of TTVR procedures are constrained by the pace of procedural dissemination and clinical adoption. To ensure that urgent clinical needs are met in a timely manner and to benefit patients worldwide as early as possible, the company has proactively invested in surgical robotics technology, driving transformation in treatment approaches through innovation.
From the continuous iteration of the LuX-Valve series to breakthroughs in the development of the iJensRobo robotic platform, Jenscare Scientific has established a comprehensive solution covering the entire treatment pathway for tricuspid valve disease, further strengthening its technological leadership in the field of structural heart disease.
Future Outlook:
Advancing Clinical Validation and Accelerating Adoption and Accessibility
We will steadily advance subsequent clinical studies of the iJensRobo robot-assisted TTVR system, accumulating more evidence-based medical data while continuously optimizing product performance. At the same time, we will expedite the regulatory approval process for iJensRobo to enable large-scale commercialization as soon as possible. Our goal is to allow more patients with urgent tricuspid regurgitation to benefit from precise and safe robot-assisted TTVR interventions, and to contribute China's innovative strength to the global field of structural heart disease treatment.
Disclaimer
The content disclosed in this article is intended for academic exchange purposes only and should not, under any circumstances, be construed as medical advice. Our company makes no representations or warranties regarding the accuracy, completeness, or timeliness of any original, reproduced, or shared content.
The data referenced herein is for informational purposes only and shall not be regarded as advice, recommendations, offers, or invitations. This material is based on publicly available information, internally developed data, and information obtained from other sources deemed reliable. However, no guarantee is made as to the absolute reliability of such information. All opinions and views expressed are based on judgments as of the date of writing and are subject to change at any time without notice.
With respect to any medical devices mentioned in this article, our company makes no representations or warranties regarding their performance or clinical outcomes in diagnostic or therapeutic applications.













